
Self-amplifying RNA (saRNA)
Revolutionizing Vaccine Development
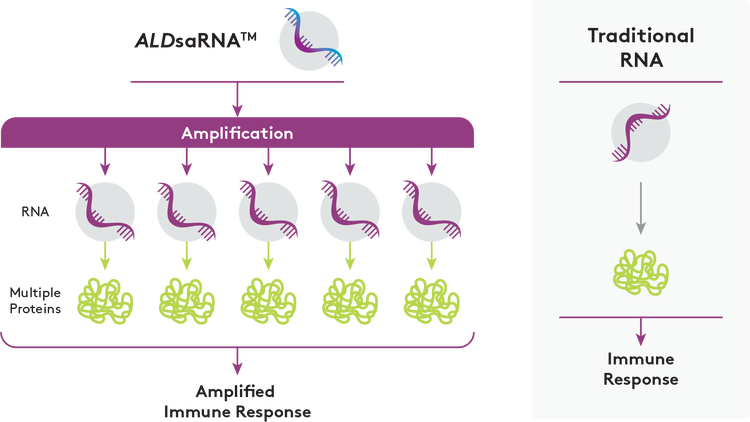
Rapid response to emerging diseases is critical when developing a vaccine. Long RNA (mRNA and saRNA) is at the forefront of vaccine development.
Benefits of saRNA
- saRNA vaccines can be developed in a fraction of the time it takes to develop a conventional vaccine
- Manufacturing is cell-free, does not require a dedicated facility
- saRNA vaccines often produce a higher immune response from a smaller dose when compared to mRNA based vaccines
Related News
Related Blog Posts
topic
mRNA