
pALD-HELP for rAAV Production
In Stock, Royalty-Free, Helper Plasmid Solution for Recombinant AAV Manufacturing
Triple transfection is the most common method for rAAV production and does not require co-infection with adenovirus; a helper plasmid provides adenoviral genes. Aldevron's pALD-HELP has several advantages:
- Immediately available at research grade and GMP-Source®
- Royalty free, including for commercial products
- Replaces any rAAV helper plasmid
- Kanamycin resistance
- Consistent, reliable supply
With Aldevron's pALD-HELP, clients do not need to manufacture a custom batch of helper plasmid because Aldevron maintains it as an in-stock product, providing a high-quality and consistent source, helping reduce timelines and cost.
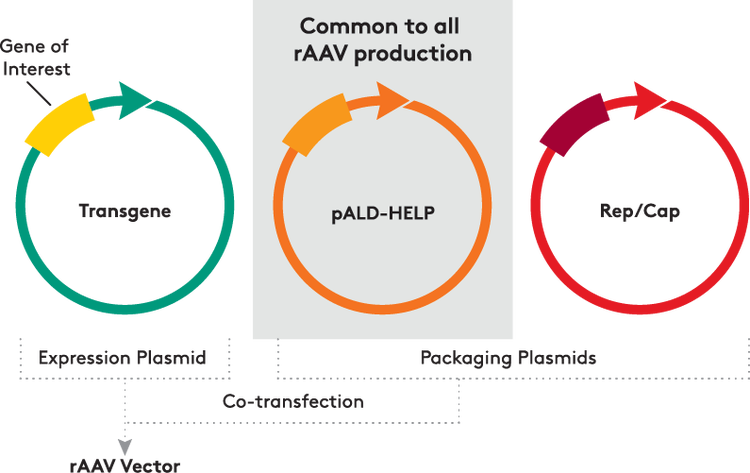
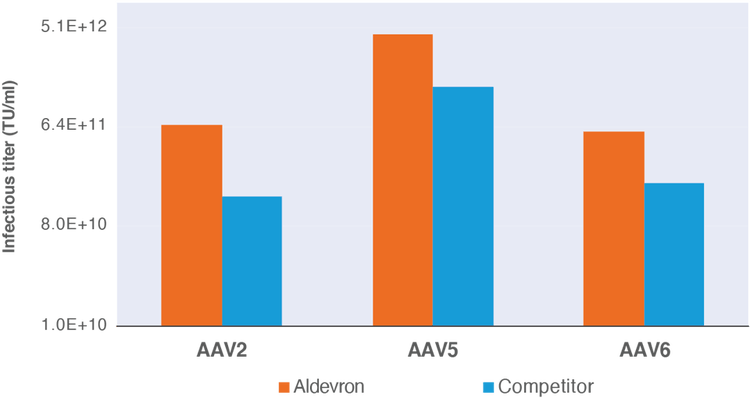
Infectious titer data for rAAV vector production in adherent HEK293T cells using pALD-HELP and pALD-AAV rep/cap plasmids as compared to commercially available rep/cap plasmids.
Ordering Information
| Product | Pack Size | Catalog Number | RG Purchase Option |
| pALD-HELP - RG | 10 mg | 5082-10 | Buy Now |
Additional discounts available for quantities > 5 vials.
| Appearance | Clear and Colorless |
| Buffer | TE |
| USP Sterility <71> * | No Growth |
| Concentration | 1.0 mg/mL +/- 10% |
| DNA Homogeneity | ≥ 80% Supercoiled |
| Endotoxin | ≤ 100 EU/mg |
| Identity | Confirmed Against DNA Ladder |
| Plasmid Identity | Identical to Reference Sequence |
| ABS 260/280 Ratio Purity | 1.80 - 2.00 |
| Residual Host Genomic DNA via qPCR | ≤ 5% |
| Residual Host Protein via BCA | ≤ 2% |
| Residual Host RNA via SYBR Gold | ≤ 5% by Semi-Quantitative Densitometry |
| Restriction Digest | Matches Expected Restriction Pattern |
| Mycoplasma Contamination | Negative for the Presence of Mycoplasma |
*Sterility testing is not available for research grade material. Bioburden by direct inoculation with a specification of no growth after 48 hours is performed at the research grade level.